CONTEC Brand New Vital Sign Patient Monitor, NIBP / SpO2 / PR CMS5100 Hot Sale
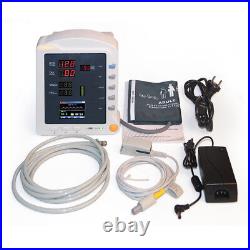
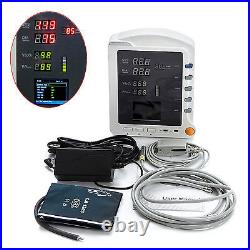

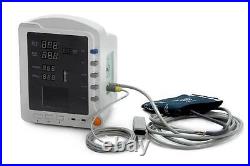







CMS5100 ICU CCU 3-Parameter Vital Sign Monitor Patient Monitor NIBP SpO2 PR. CMS5100 Patient Monitor is a feature-rich monitoring device that can be applied to adults, pediatric and neonate.
User can choose different measurement parameters according to different needs. The patient monitor can monitor NIBP, SpO2 and PR. It integrates the function of parameter measuring and displaying into a compact, lightweight patient monitor, which is suitable for all levels of hospitals, community medical and home use. 1Be applicable for NIBP and SpO2 monitoring of adult, pediatric and neonate of all ages, easy to operate, high cost-effective.
2Be applicable for medicine, surgery, operating room, ICU/CCU, emergency room, obstetrics and gynecology, pediatrics. 3Compact and flexible appearance, easy for carrying and suitable for indoor and outdoor (in ambulance) monitoring.4Built-in rechargeable lithium polymer battery, ensuring uninterrupted monitoring. 5Perfect menu design, user-friendly interface. 6Display the measurement result of SYS, DIA, MAP, SpO2, PR and bar graph by the display screen of high-brightness digital tube. 72.8'' (320×240) true color TFT LCD screen, displays the information of time, SpO2 Plethysmogram, alarm condition, trend graph, list and system settings, etc.
8Visual and audible alarm for SYS, DIA, MAP, SpO2 and PR, and upper and lower limit of alarm can be set as necessary. 9Independent nonvolatile memory, storage for up to 2,000 groups of NIBP data and 78,000 groups of SpO2 data.
10Convenient and quick in reviewing measurement data, available for reviewing the NIBP trend graph of 24 hours and SpO2 trend graph of 22 hours. Using Oscillometry technology, also called Oscillography technology to measure the blood pressure. 2Measurement interval in auto mode: 1, 2, 3, 4, 5, 5×n(n=2, 3, 4...51) min. 4Accuracy: max mean error: ±5 mmHg; max standard deviation: 8 mmHg.5Overpressure protection: dual protection for both software and hardware. 6Others: reset, self-testing and accuracy testing of static pressure. Using Photoelectric Oxyhemoglobin Inspection Technology combined with Capacity Pulse Scanning & Recording Technology to measure the saturation of blood oxygen. 1Measurement range: 0 %100 %.
2Alarm range: 0 %100 %. 4Accuracy: 70 %100 %, ±2 %; below 70 % unspecified. Using Photoelectric Oxyhemoglobin Inspection Technology combined with Capacity Pulse Scanning & Recording Technology to measure the pulse rate. 1Measurement range: 25 bpm250 bpm. 2Alarm range: 25 bpm250 bpm.4Accuracy: ±2 bpm or ±2 %, whichever is greater. 4 Safety and Power supply. 1Power supply: rated voltage: 220 V, rated power: 50 Hz. 2Safety classification: Class I equipment, type BF defibrillation-proof applied part. Dimension: 190 mm(L)×162 mm(W)×240 mm(H).
Weight: about 1.6 kg. Food and Drug Administration and state and local regulatory agencies. The Fingertip Pulse Oximeter is registered on the Australian Register of Therapeutic Goods (ARTG)with the code 197923, and certified by FDA of United States and CE, TUV of Europe. The Fingertip Pulse Oximeter that is FDA 510K Approved. These charges are buyers' responsibility. We are looking for partners in the world now! Do you want to become our agent in you conutry?This item is in the category "Business & Industrial\Healthcare, Lab & Dental\Medical & Lab Equipment, Devices\Patient Monitors". The seller is "contecmedical1826" and is located in this country: CN. This item can be shipped worldwide.
- Model: CMS5100
- Featured Refinements: Vital Signs Monitor
- Features: Battery Operated
- MPN: 69450401
- Brand: Con tec